Because of their unique structure, biological tissues exhibit physical and mechanical properties that are unlike anything in the world of engineering.
Because of their unique structure, biological tissues exhibit physical and mechanical properties that are unlike anything in the world of engineering.
So, by learning from nature, novel engineering systems might be developed that utilise the principles found in natural materials – a field of technology that has been termed biomimetics.
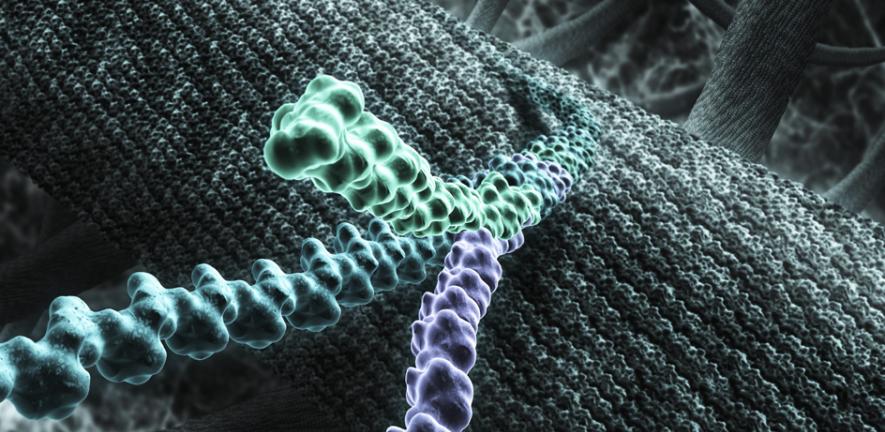
Mention the word ‘biology’ and most people think immediately about cells. However, large portions of the human body are non-cellular and are made instead from an extracellular matrix (ECM) that provides much of the structural support around cells. This supportive function of the ECM is especially evident in the connective tissues of the body. Many load-bearing structures such as bones, teeth and ligaments are connective tissues and these have been the focus of recent bioengineering research in Cambridge.
Dr Michelle Oyen, Lecturer in the new Engineering for Life Sciences programme in the Department of Engineering, is studying the mechanical functions of connective tissues. Her research ranges from fundamental science and engineering projects through to collaborative projects with clinicians for developing mechanics-based tools for use in medical practice. The unifying theme of this research lies with the primary component of many connective tissues: the structural protein collagen.
Building blocks of natural materials
Collagen is ubiquitous; this triple-helical protein makes up a quarter of all proteins in the body. It self-assembles from the molecular scale up to large fibre-like structures, creating a hierarchical material with remarkable physical properties. Collagen combines with other ECM components – mainly water, non-collagenous proteins and sugars – and, in mineralised tissues, with bioceramics analogous to earth minerals. These non-living, but cell-derived, materials combine with cells to form living yet mechanically robust tissues.
Collagen takes on different roles in different parts of the body. In structural tissues, like bones and ligaments, it’s found in rope-like fibres that provide resistance to stretching and tearing forces. In cartilage, which is mostly loaded in compression, collagen has more of a ‘holding’ function, with the fibres arranged rather like a basket, retaining other hydrated proteins and sugars. In the lens of the eye, collagen is crystalline, organised precisely for optical transparency. In fact, there are over 20 different types of collagen in the body, and it is not even known precisely what functions they all fulfil.
Mechanics in medicine
The study of the biomechanical properties of collagen and ECM is a particularly exciting and fast-growing field in reproductive medicine. One aspect of Dr Oyen’s research has been to examine the physical properties of the ECM in the amniotic sac, the membrane that ruptures (the ‘breaking of waters’), signalling imminent birth.
Rupture occurring before full-term gestation results in approximately a third of all premature births. Following the first-ever set of rigorous bioengineering studies on placental membranes, Dr Oyen and clinical colleagues at the University of Minnesota concluded that the phenomenon is due to localised damage, not widespread overall membrane deterioration, and that diagnostic techniques may be developed to detect localised thinning and ECM damage for intervention into premature birth.
This project is taking a new direction since Dr Oyen’s arrival in Cambridge. By teaming up with researchers at the newly opened Centre for Trophoblast Research (www.trophoblast.cam.ac.uk), she will be able to examine placental development from an engineering and mechanics perspective.
Mimicking nature
Nature clearly creates dynamic, mechanically functional tissues that are different from anything engineers have made. As an example, cartilage, which forms the gliding surface that permits joint movement, is approximately 75% water and only 25% collagen, sugar and other proteins, and yet its stiffness and shock-absorbing capability make it comparable to solid rubber. Moreover, the cartilage-on-cartilage sliding interface has lower friction than ice sliding on ice.
In fact, when engineers design materials, uniformity and simplicity are often prized. Engineering materials do not always feature the multi-level hierarchical organisations found in protein-based materials, nor do they exemplify the dramatic spatial non-uniformity that has been found to strengthen natural materials. So, by learning from nature, novel engineering systems might be developed that utilise the principles found in natural materials – a field of technology that has been termed biomimetics.
In some instances, biomimetics takes the form of direct imitation, as in the case of a nanocomposite of mineral and proteins similar to natural bone. For cases of major bone defects, such as occurs through trauma or cancer, a bone-like material that is biocompatible can be seeded with cells to form a ‘tissue-engineered’ construct and implanted within the body. However, if you consider just how lightweight, yet stiff, strong and tough, a bone-like material is, why not use it for other structural applications such as architecture? This is a challenge that Dr Oyen is investigating.
To do this, you need to go back to first principles – how the material forms. With funding from the Royal Society, Dr Oyen is examining biomineralisation and the formation of mechanically robust bone-like materials. The work differs from tissue-engineering approaches in that there is no cellular component and the end applications are viewed as being remote from medicine. Although a large number of groups have considered the synthesis of biomimetic materials, far fewer have taken a primary angle associated with the measurement of mechanical properties. Dr Oyen views the materials as successful when they replicate both bone composition and mechanical behaviour.
Inspirational materials
It is also possible to abstract ideas from nature without directly imitating the materials themselves. As examples, key concepts that could guide the formation of ‘bone-inspired’ materials include: composite materials with a very large stiffness mismatch between the phases; materials that form from room-temperature deposition of a ceramic onto a self-assembled polymer; materials with up to seven different levels of hierarchical organisation; and materials that are self-healing. Each of these concepts could be applied to a system that is not protein based, and ongoing research both at Cambridge and across the world is incorporating these types of principles for materials development.
Compared with many branches of engineering, biomimetic engineering is comparatively new. In this rapidly expanding field, the lessons learnt from the physical and mechanical properties of natural materials such as collagen and bone offer great promise within an engineering framework. Not only is this sure to make a difference to 21st-century healthcare, but there are also ways in which engineering will itself benefit from the abstraction of ideas from nature.
For more information, please contact the author Dr Michelle Oyen (mlo29@cam.ac.uk)
at the Department of Engineering.
This work is licensed under a Creative Commons Licence. If you use this content on your site please link back to this page.





