Bacteria 'plan ahead' by tightening their belts to help them survive looming lean periods, researchers at Cambridge have discovered.
Bacteria 'plan ahead' by tightening their belts to help them survive looming lean periods, researchers at Cambridge have discovered.
If we can understand how bacteria use signalling systems to cope with the drugs we’re throwing at them, that should help us find new ways to get around their defences
Hannah Gaimster
At a time when there is growing concern about rising antibiotic resistance, the results – published in the journal PLOS ONE – could lead to new ways of combating dangerous bacteria.
For many years, people thought bacteria worked by simply consuming resources until they ran out, and then entering a stationary, non-growing, phase. Working with physicists from the Cavendish Laboratory, Hannah Gaimster and David Summers from the Department of Genetics showed that as resources decline, bacteria switch to austerity mode, individually deciding to consume less until conditions improve.
Summers and Gaimster identified this behaviour by studying indole – the chemical behind the bad smell in babies’ nappies (and also used as an ingredient in perfumes) – a key signalling molecule in bacteria.
Scientists have known for many years that bacteria use low concentrations of indole to communicate with each other. This study shows for the first time that bacteria also use the chemical in a completely different and dramatic way – producing high transient pulses of indole that accumulate within a bacterial cell and cause it to enter austerity mode.
According to Gaimster: “The effects of low concentration of indole are subtle, but higher concentrations will do things such as abolish the ability of the cells to divide, or shut down growth completely.”
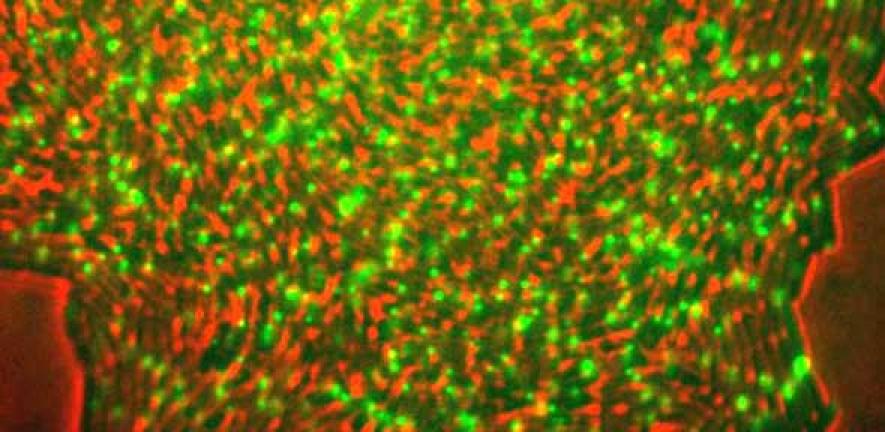
Working with Escherichia coli, Gaimster and Summers grew two cultures of the bacteria, one was the normal indole-producing E. coli and the other a mutant strain that has lost its ability to make indole.
When they stopped feeding the bacteria for 10 days, they found that the two strains coped with famine in very different ways.
“Initially the mutants seemed to do better than the wild type bacteria, but when we looked at the longer term, over 10 days the wild type’s viability declined by only one third compared with the mutants, whose viability fell by 80%. This is because without indole, the mutants could not plan ahead and weren’t able to survive as well in the long-term,” she explained.
In a second experiment, they seeded a population of wild type E. coli with 1% of the indole-lacking mutants and filmed what occurred when famine set in.
“When the food began to run out, the normal cells started doing this careful slowing down to make the food last longer, but the mutants ran riot through the culture, increasing many fold in number and eating all the food reserves. That results in disaster for everyone, because they all starve to death,” Gaimster said.
“Our results have many applications because indole plays such an important role in how bacteria respond to stress, including antibiotics. If we can understand how they use signalling systems to cope with the drugs we’re throwing at them, that should help us find new ways to get around their defences,” she said.
By discovering an entirely new way that bacteria use indole to communicate, the study could help scientists find new ways of controlling dangerous bacteria such as MRSA.
As well as opening up new modes of attacking dangerous bacteria, the study illustrates that rather than the simple creatures we assume them to be, bacteria are complex organisms with sophisticated survival strategies.
According to Summers: “I want to destroy the idea that bacteria are simple, because that’s an extraordinarily naïve view of a very sophisticated cell. Compared with humans, bacteria have been evolving for an unbelievably long time, so they will be better at being bacteria than any other organism will be at being itself.”
This work is licensed under a Creative Commons Licence. If you use this content on your site please link back to this page.