The Cambridge Animal Alphabet series celebrates Cambridge's connections with animals through literature, art, science and society. Here, F is for Fruit Fly and the myriad ways that they are helping with medical research.
The Cambridge Animal Alphabet series celebrates Cambridge's connections with animals through literature, art, science and society. Here, F is for Fruit Fly and the myriad ways that they are helping with medical research.
More is known about the biology of Drosophila than possibly any other animal on earth. I expect that Drosophila will remain a vital model organism for many decades to come.
Simon Collier
Scroll to the end of the article to listen to the podcast.
Each morning a yeasty smell drifts through the basement of the Genetics Building. Research technician Huai Xue Lin arrives early to cook the food needed for millions of fruit flies. The Drosophila is not a picky eater: it thrives on a mix of cornmeal, sugar and yeast, mixed with agar to make it solid. The fly kitchen operates an impressive takeaway service, supplying not just the Fly Lab on the first floor but also Drosophila research facilities all over Cambridge.
As their name suggests, fruit flies are the small insects that appear on hot summer days to feast on the surface of ripening fruit – or sup on any wine or beer left out. Once Drosophila detect something sweet and sticky, they are annoyingly persistent but they pose no threat to human health.
Fruit flies are used by research groups throughout Cambridge to learn more about how genes determine development. That’s because, despite looking remarkably dissimilar to us, Drosophila have much the same fundamental biological make-up as humans. Significantly for medical scientists, they share 75% of the genes that cause disease in the human population.

Drosophila are not hard to raise in huge numbers. Their eggs hatch within 24 hours. The larva that crawls out eats and grows non-stop for about four days. It then pupates for around four days before emerging as an adult fly. The fly is sexually mature and ready to mate within a few hours.
Because fruit flies reproduce so fast, researchers use them to track ways in which traits, including genetic abnormalities, are transferred down many generations in a relatively short time. Drosophila are also easy to anaesthetise using carbon dioxide – and make a speedy recovery. These characteristics combine to make the fruit fly a valuable model for research into genetics and associated fields.
The potential offered by Drosophila as a tool for understanding the principles of heredity was first explored in the USA early in the 20th century when Thomas Hunt Morgan won the Nobel Prize "for his discoveries concerning the role played by the chromosome in heredity". British scientists began to use fruit flies as a research organism after the Second World War with the first fruit fly facility established in Cambridge in the 1960s.
Professor Michael Ashburner recalls the early days of Drosophila research in Cambridge: the flies were reared in milk bottles in a temporary lab located in suburban Cambridge. Ashburner and colleagues carried out extensive fundamental work to determine how genes control complex traits such as height and weight. He went on to become a pioneer in the use of computing in biology, developing a standardised vocabulary that enables scientists’ observations to be read by a computer.
As a key player in the sequencing of Drosophila by a public-private consortium, Ashburner helped to ensure that the research was made publicly available. His book Won for All: How the Drosophila Genome Was Sequenced is a compelling account of the highs and lows involved in a hugely ambitious project involving a number of institutions.
Today the Fly Lab is a modern facility on the first floor of the Genetics Building. Along one wall are storage units housing thousands of tubes containing live fruit flies. The tubes provide a ‘library’ of ‘stocks’ with each stock relating to a particular research project. The Lab is equipped with 24 work stations for researchers working on aspects of genetics. In addition, batches of Drosophila are also supplied to research groups working elsewhere in Cambridge.
The fields covered range from neurodegenerative disease to parasite interactions. The most recent addition to the Lab’s clients is the Hannon Group at Cambridge Biomedical Campus which is using Drosophila as one of many pathways for developing new methods for cancer diagnosis, treatment and prevention.
Fly Lab manager, Dr Simon Collier, says: “Flies can be used to address a wide variety of problems in biology and medicine. The Fly Lab provides a resource not just to fly workers in Cambridge but elsewhere in the UK and Europe. I believe we can be especially helpful to research groups that are largely clinical but also want to incorporate the fly model into their research.”
A quick look at the website FlyBase gives a picture of the myriad ways in which the humble fruit fly is contributing to medical science. The Cambridge branch of FlyBase is headed by Professor Nick Brown who also leads a research lab in the Gurdon Institute.
The Brown Lab uses Drosophila to investigate how bodies are built and how, during the development of an organism, cells attach to each other by means of ‘cell adhesion’. The processes which determine the growth of an adult organism from a single cell, the fertilised egg, are extremely complex and involve receptors known as ‘integrins’. By understanding the ways in which ‘faults’ can occur in fruit flies, the group will be able to contribute to the development of treatments for human conditions such as skin blistering diseases, muscular dystrophies and aberrant blood clotting.
A lab headed by Professor Steve Russell is investigating the genes that control the activity of other genes, particularly a group called ‘Sox genes’. These types of genes are important in both humans and flies as they often control the behaviour of tissues by regulating the particular set of genes active in each cell. The Russell lab is looking particularly at development of the central nervous system and gonads. These are just two of many groups using Drosophila as a model in research.
A side room at the Fly Lab is set aside for Sang Chan, the Lab’s Microinjection Specialist. He injects fly eggs with DNA so that researchers can produce gene mutations in flies that will enable them to track the functional effects of genes – and thus identify the genes regulating the production of particular protein configurations or the behaviour of other genes.
Looking through a powerful microscope, he uses an instrument called a ‘micromanipulator’ to push a needle into a fly egg about 0.5mm in length (roughly the size of a coarse grain of sand) in order to inject DNA roughly equivalent in volume to a millionth of a drop of water. It took Chan six months to acquire the fine motor skills needed to carry out this delicate task with reliable accuracy.
Running the Fly Lab is a round-the-clock enterprise that requires constant attention to detail. The storage units are kept at a constant temperature of 25 degrees, the temperature at which Drosophila thrive best. Double doors and other precautions prevent flies from escaping: many are transgenic and, as such, considered a potential ecological hazard. The Lab has strict hygiene regulations designed to keep the presence of mites (which live on fruit flies) to a minimum.
“More is known about the biology of Drosophila than possibly any other animal on earth. For this reason alone, I expect that Drosophila will remain a vital model organism for many decades to come. The short generation time, relatively simple genome and ease of culture are as useful today as they were in Thomas Morgan’s time,” says Collier. “Our increasingly molecular and cellular perspective on human disease has brought medical research to a level where humans and flies are understood to be remarkably similar and means the fly can be an effective model for human disease.”
Next in the Cambridge Animal Alphabet: G is for the world's second fastest animal, which flanks the escutcheons of King's College Chapel and is playing an important role in research into treatments for osteosarcoma.
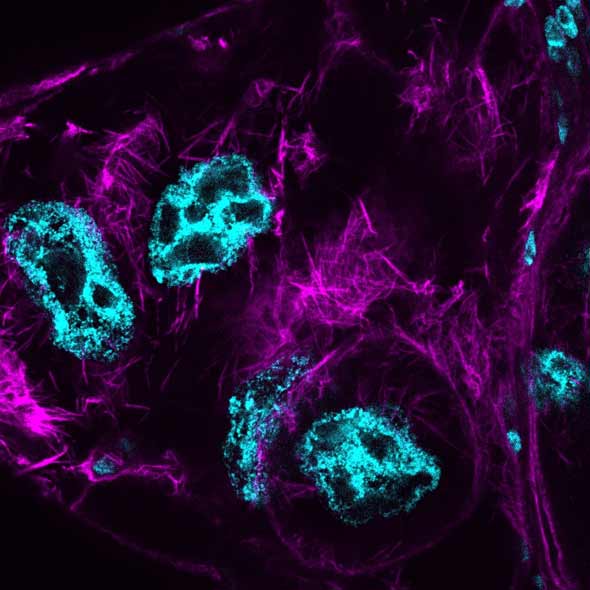
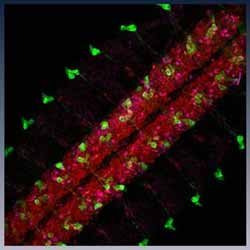
Inset images: D. melanogaster dorsal open wings (Sylwester Chyb and Nicolas Gompel); Actin cables in Drosophila nurse cells during late-oogenesis. At this stage, nurse cells die and extrude their cytoplasm into the developing oocyte. This process is required for viable eggs to develop. Cyan = DNA (DAPI), highlighting the nuclei; Magenta = Actin (Phalloidin), highlighting enrichments of Actin that form across the cells (Tim Weil and Anna York-Andersen, Weil Lab); Drosophila embryo - the large stripe that you see along the centre of the embryo is the developing nervous system and subsets of neurones have been labelled in green (Holly Ironfield and Eva Higginbotham).

The text in this work is licensed under a Creative Commons Attribution 4.0 International License. For image use please see separate credits above.