Cambridge alumnus Richard Henderson (Corpus Christi College, 1966) has been jointly awarded the 2017 Nobel Prize in Chemistry, along with former Cambridge University senior research associate Joachim Frank, and Jacques Dubochet from the University of Lausanne, Switzerland.
Cambridge alumnus Richard Henderson (Corpus Christi College, 1966) has been jointly awarded the 2017 Nobel Prize in Chemistry, along with former Cambridge University senior research associate Joachim Frank, and Jacques Dubochet from the University of Lausanne, Switzerland.
Henderson completed his PhD in 1970, carrying out his research under the supervision of David Blow at the Medical Research Council Laboratory of Molecular Biology, where he is currently based. He is an Emeritus Fellow of Darwin College and an Honorary Fellow of Corpus Christi.
Frank, now based at Columbia University, New York, USA, was a senior research associate at Cambridge’s Cavendish Laboratory from 1973-1975.
The three researchers have received the award "for developing cryo-electron microscopy for the high-resolution structure determination of biomolecules in solution". According to the Nobel Committee, this method “has moved biochemistry into a new era”.
Professor Ben Luisi, from Cambridge's Department of Biochemistry and who led a consortium to establish a cryo-electron microscopy facility at the University, said: "The award recognises and celebrates the development and wonderfully successful application of electron microscopy to visualise isolated biological molecules in sufficient detail to infer the relative positions of the constituent atoms or to find the locations of the molecules in large complexes within cells.
"This approach has tremendously expanded the range of biological samples that can be studied. It has enabled deeper understanding of how biological molecules recognise each other to form complex organisations, how they make and break chemical bonds, how they work like motors, and how they communicate information through structural change.
"The methods have taken decades to develop and were driven and sustained by Richard's deep theoretical insights and vision. The molecular biology community has benefited tremendously from the work of Richard and his colleagues. The university has also benefited from Richard's generous help and advice to establish electron microscopy facilities here."
Dr Luca Pellegrini, also from the Department of Biochemistry, said: "We’re delighted about the news that the 2017 Nobel prize in Chemistry was jointly awarded to Dr Richard Henderson of the MRC Laboratory of Molecular Biology in Cambridge. The award recognises Dr Henderson’s long-standing interest in electron microscopy and its application to fundamental biological problems.
"The pioneering research carried out by Dr Henderson in the field of electron microscopy has revolutionised the structural investigation of biological specimens under native conditions, leading to a major breakthrough in our ability to obtain high-resolution images of macromolecular assemblies of biological and medical interest."
Professor Magdalena Zernicka-Goetz, from the Department of Physiology, Development and Neuroscience, said: “I think it is wonderful. A visual image is the essential component to understanding, often the first one to open our eyes, and so our minds, to a scientific breakthrough.”
This brings the total number of affiliates of the University of Cambridge who have been awarded the Nobel Prize to 98.
Cool microscope technology revolutionises biochemistry
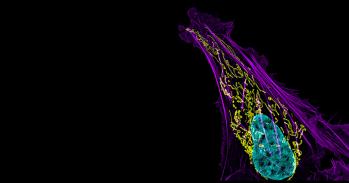
A picture is a key to understanding. Scientific breakthroughs often build upon the successful visualisation of objects invisible to the human eye. However, biochemical maps have long been filled with blank spaces because the available technology has had difficulty generating images of much of life’s molecular machinery. Cryo-electron microscopy changes all of this. Researchers can now freeze biomolecules mid-movement and visualise processes they have never previously seen, which is decisive for both the basic understanding of life’s chemistry and for the development of pharmaceuticals.
Electron microscopes were long believed to only be suitable for imaging dead matter, because the powerful electron beam destroys biological material. But in 1990, Richard Henderson succeeded in using an electron microscope to generate a three-dimensional image of a protein at atomic resolution. This breakthrough proved the technology’s potential.
Joachim Frank made the technology generally applicable. Between 1975 and 1986 he developed an image processing method in which the electron microscope’s fuzzy twodimensional images are analysed and merged to reveal a sharp three-dimensional structure.
Jacques Dubochet added water to electron microscopy. Liquid water evaporates in the electron microscope’s vacuum, which makes the biomolecules collapse. In the early 1980s, Dubochet succeeded in vitrifying water – he cooled water so rapidly that it solidified in its liquid form around a biological sample, allowing the biomolecules to retain their natural shape even in a vacuum.
Following these discoveries, the electron microscope’s every nut and bolt have been optimised. The desired atomic resolution was reached in 2013, and researchers can now routinely produce three-dimensional structures of biomolecules. In the past few years, scientific literature has been filled with images of everything from proteins that cause antibiotic resistance, to the surface of the Zika virus. Biochemistry is now facing an explosive development and is all set for an exciting future.
Information taken from a press release from the Royal Swedish Academy of Sciences.

The text in this work is licensed under a Creative Commons Attribution 4.0 International License. For image use please see separate credits above.