Adult skates can spontaneously repair cartilage injuries

Researchers have found that adult skates have the ability to spontaneously repair injured cartilage, using a type of cartilage stem cell. Human cartilage has very limited capacity for repair, and the finding may lead to new stem cell treatments for human cartilage injuries.
Published today in the journal eLife, the study identified a new type of cartilage stem cell in the skeleton of adult skates, Leucoraja erinacea. These cells enable skates to keep making new cartilage throughout their life, so their skeleton can keep growing and any cartilage injuries can be repaired.
Current stem cell therapies for cartilage repair in humans are not very effective because lab-engineered cartilage from adult stem cells has a tendency to start turning into bone. Previously, no animal has been found to have the ability to make new cartilage during adulthood that stays as cartilage, rather than turning into bone, or have the ability to spontaneously repair injured cartilage.
“Cartilage injury in humans – for example because of osteoarthritis or a sports injury – is a huge problem, and a huge economic burden,” said Dr Andrew Gillis from the University of Cambridge’s Department of Zoology and the Marine Biological Laboratory in Woods Hole, USA, who led the research.
“We are tremendously excited to find that skates can spontaneously repair injured cartilage, and to have an insight into how this happens. It paves the way for developing better treatments to repair cartilage injuries in humans, which are currently very limited in their effectiveness.”
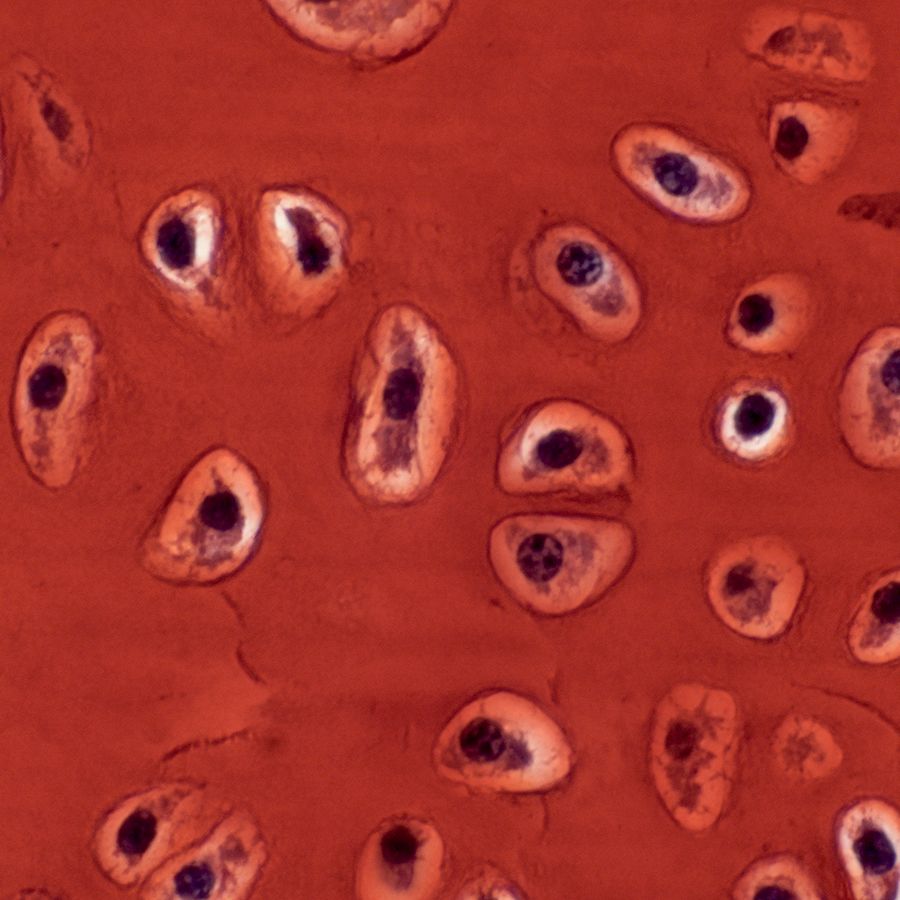
Cartilage cells within the fin cartilage of a skate hatchling
Cartilage cells within the fin cartilage of a skate hatchling
The cartilage stem cells, called chondroprogenitors, were found in the fibrous tissue, called the perichondrium, which wraps around cartilage in the adult skate. By labelling these stem cells with a fluorescent marker, the researchers could trace the cells they created - and found that they ended up as cartilage in the adult skeleton. These cells were also shown to express genes that control cartilage development.

Fin cartilage of a skate embryo, showing expression of the gene encoding collagen (green) in cartilage cells
Fin cartilage of a skate embryo, showing expression of the gene encoding collagen (green) in cartilage cells
This study found that embryonic development of cartilage in the skate closely mirrors that in humans, but unique features of the adult skate skeleton – including the presence of cartilage stem cells in the perichondrium – facilitate the continued growth of cartilage throughout life.
Most cartilage in humans is formed as the skeleton first develops, and this is later replaced by bone. By the time adulthood is reached, cartilage only exists in a few places in the body such as the joints. Human cartilage has no blood or nerve supply, and it has no resident stem cell population, so it has very limited capacity for repair.
Osteoarthritis is a debilitating deterioration of joint cartilage, with symptoms ranging from stiffness and joint pain to complete immobility. It can severely impact quality of life, and has an extremely high economic burden, so there is great interest in identifying novel therapeutic strategies to promote joint cartilage repair.
The researchers warn that taking ‘shark cartilage’ tablets should not be considered as a cure for joint pain or any other illness, as there is no scientific evidence that they work.
Their next step is to understand the molecular mechanisms that allow this specialised stem cell type to make cartilage as a stable tissue in adult skate. They hope this will allow them to manipulate human stem cells to behave in a similar way, and enhance their ability to generate stable cartilage for transplantation and repair.
This research was funded by Wellcome, the Royal Society, the Isaac Newton Trust and the Fisheries Society of the British Isles.
Reference: Marconi, A. et al: ‘Adult chondrogenesis and spontaneous cartilage repair in the skate, Leucoraja erinacea’, ELife, May 2020, DOI: 10.7554/eLife.53414
Image credits: Andrew Gillis